THE HARD SCIENCE
BEHIND WHY COPPER WORKS
COPPER and COPPER ALLOYS as ANTIMICROBIALS
Many Metals Cause Death of Microbes (Mercury, Silver, Bismuth, Copper….). Copper is unique. In its solid form, it is an effective antimicrobial in reasonable time frames while being physically inert to mammals. It is an essential micronutrient (United States Recommended Daily Allowance of 0.9 mg/day) ingested as copper ions as part of a normal diet or supplement. In its un-ionized solid forms, copper is durable and wear resistant. As such, it can be formed into various useful configurations while maintaining its antimicrobial properties.
While our ancestors may not have related “antimicrobial” activity to their use of copper, they realized that water held or carried in buckets made of copper or copper alloys was not slimy when compared to water held or carried in buckets made of wood. Copper piping provided water of greater clarity and less odor than many other piping materials. And copper has been/is used in roof construction or placed in strips at the higher level rows of roofing shingles to reduce or eliminate mold growth on the roofs. Copper is also used, particularly in brass form, to prevent bio-marine fouling.
- Enterobacter aerogenes
- Staphylococcus aureus
- Pseudomonas Aerugnosa
- Drug Resistant Staphylococcus aureus
- Clostridium difficile
- Influenza A virus
- Adenovirus
- Fungi
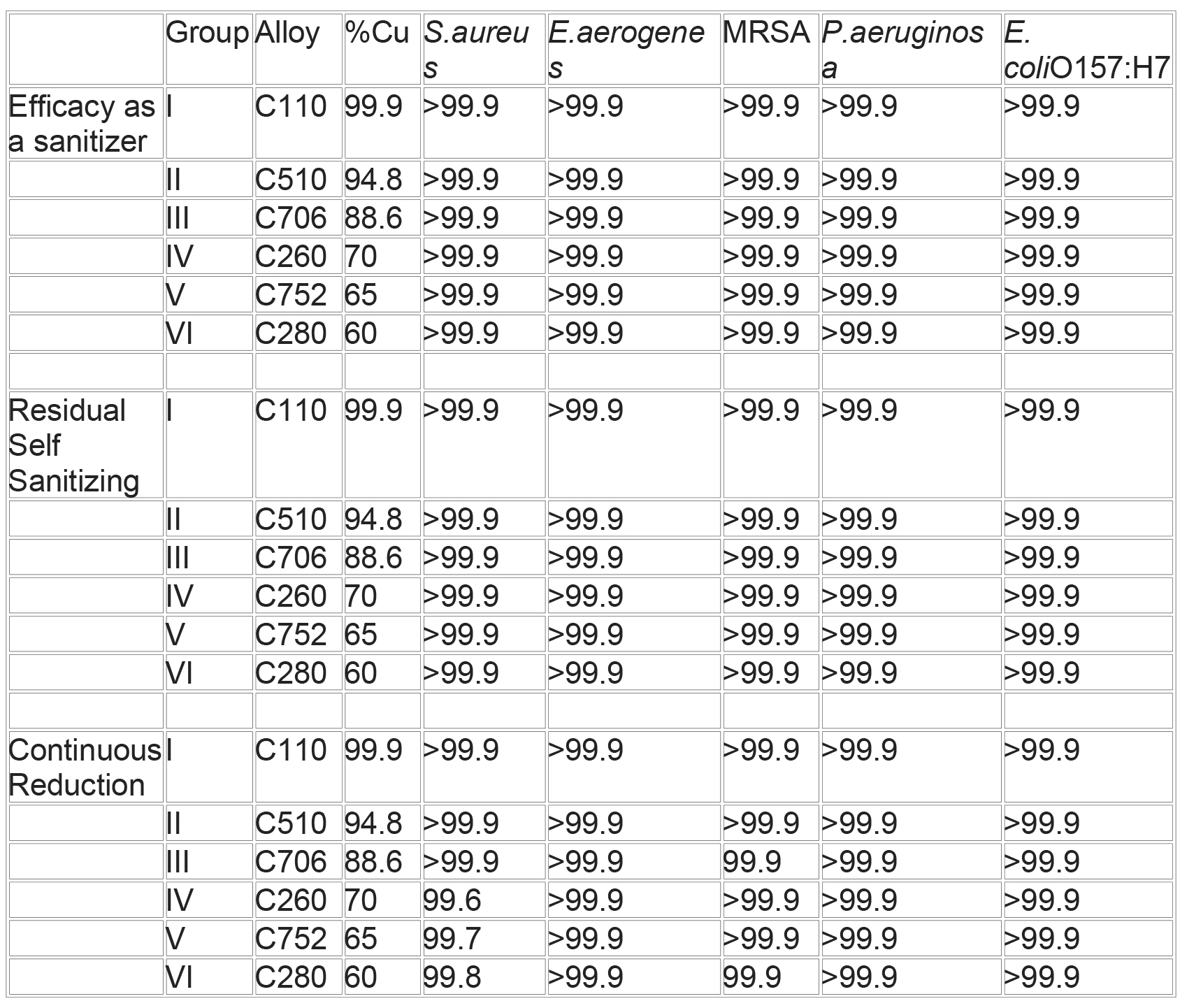
Effectiveness of Microbial Kill with Solid Copper
The reduction seen in the remaining nine tests ranged from 99.3% to 99.9%. These results indicate that the antimicrobial response of copper alloys is effective, enduring and reproducible.
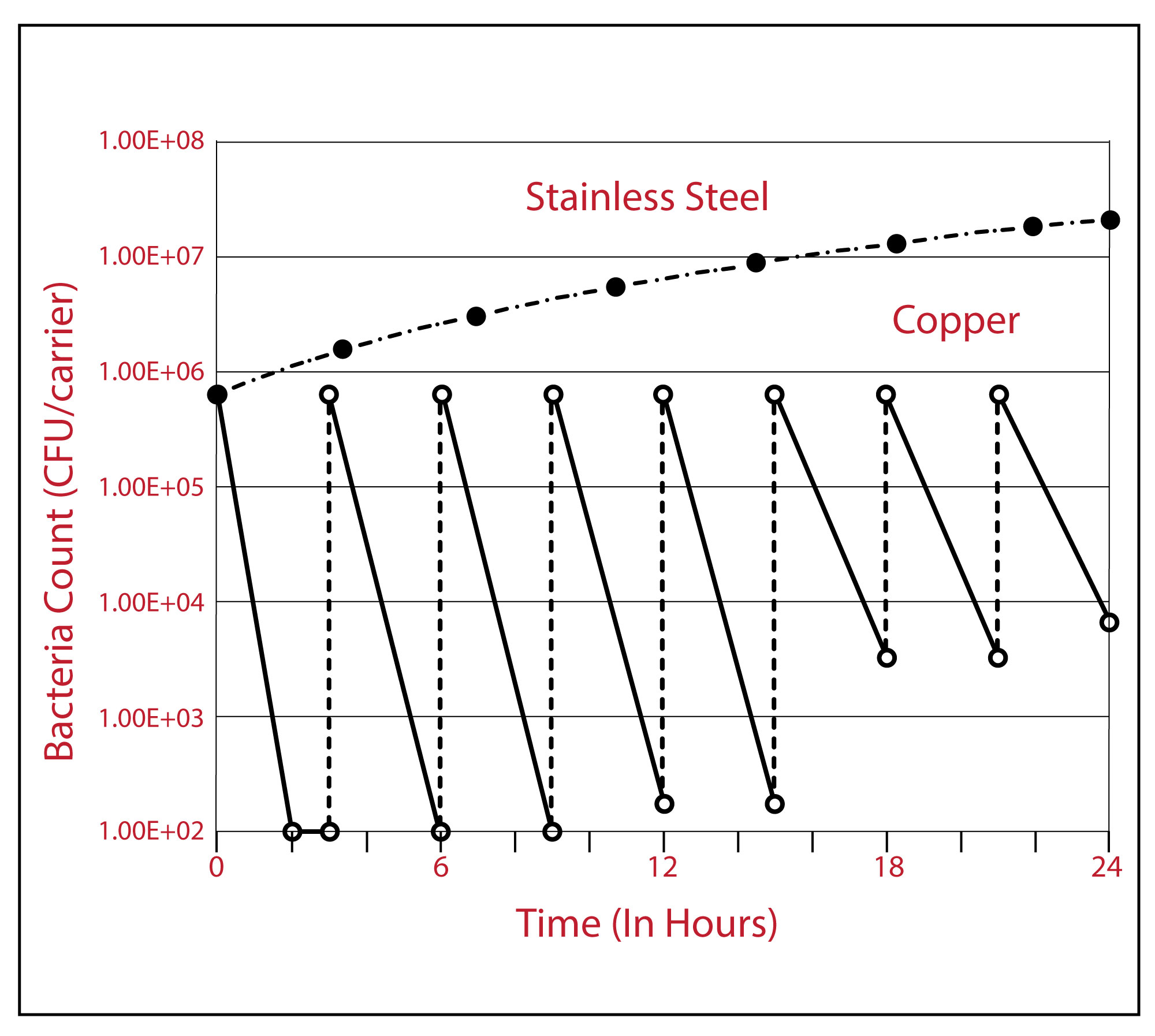
Continuous Kill Effect of Copper on Microbes
Figure 2: Continuous Reduction test results for MRSA on copper alloy C11000 and stainless steel S30400. Each inoculation adds 650,000 CFUs.
OBSERVATION: This shows the self regenerative antimicrobial power of the copper surface. If someone uses it on their hands, then puts it in their pocket, the surface will be microbe free in a very short time.
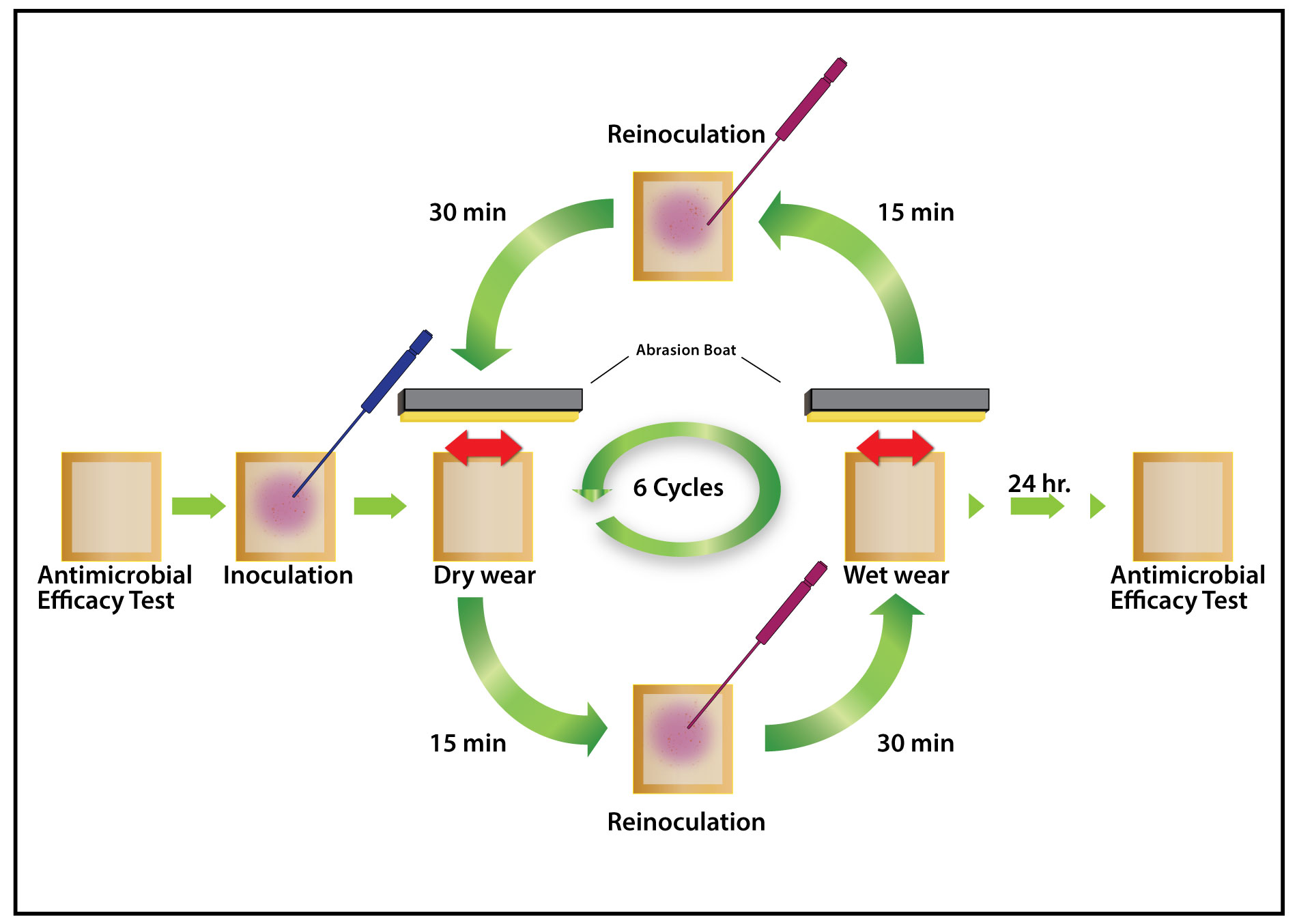
Dry Kill of Microbes vs Wet Kill of Microbes on Copper
Espírito Santo C, Lam EW, Elowsky CG, Quaranta D, Domaille DW, Chang CJ, Grass GAppl Environ Microbiol. 2011 Feb; 77(3):794-802.
Kill Rate of Microbes Versus Temperature
NOTE that the kill time decreases by 1/3 going from 4 °C (39 °F) to 20 °C (68 °F). Going to 37 °C (98.6 °F) should decrease by 1/3 again, but a reference to research at body temperature has not been found.
Wilks, SA; Michels, H; Keevil, CW (2005). “The survival of Escherichia coli O157 on a range of metal surfaces”. International Journal of Food Microbiology
(PMID) (PMID:16253366) (PMID:doi10.1016/j.ijfoodmicro.2005.04.021)
Michels, H. T.; Wilks, S. A.; Noyce, J. O.; Keevil, C. W. 2005, Copper Alloys for Human Infectious Disease Control, Presented at Materials Science and Technology Conference, September 25–28, 2005, Pittsburgh, PA; Copper for the 21st Century Symposium.
Other Information on the Kill Effects of Solid Copper/Copper Alloys The study of the antimicrobial properties of metallic copper surfaces is a relatively recent development and gained momentum when the Environmental Protection Agency (EPA) registered almost 300 different copper surfaces as antimicrobial in 2008.
https://www.antimicrobialcopper.org/epa-registration
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3067274/ )
For the most part, the bacterial kill rate of copper alloys increased with increasing copper content of the alloy. This is further evidence of copper’s intrinsic antibacterial properties.
Michels, H. T.; Wilks, S. A.; Keevil, C. W. (2003), The Antimicrobial Effects of Copper Alloy Surfaces on the Bacterium E. coli O157:H7, Proceedings of Copper 2003 – Cobre 2003, The 5th International Conference, Santiago, Chile, Vol. 1 – Plenary Lectures, Economics and Applications of Copper, pp. 439–450, The Canadian Institute of Mining, Metallurgy and Petroleum, Montreal, Quebec, Canada, (presented in Santiago, Chile, November 30–December 3, 2003)
Updated Draft Protocol for the Evaluation of Bactericidal Activity of Hard, Non-porous Copper Containing Surface Products issued on 1/29/2016. Amongst the requirements listed is “An effective product is expected to achieve a 3 log10 reduction (LR) in viable bacteria (compared to the stainless steel control) for each microbe within a 1 hr contact period.”
(copper_and_copper-alloy_surface_protocol_revised_012916)
MRSA and VRSA
VRSA (staph) is a strain of Staphylococcus aureus that is resistant to the antibiotic called vancomycin. The acronym, VRSA, stands for vancomycin-resistant Staphylococcus aureus. VRSA can cause an illness from skin infections to severe invasive disease which can result in pneumonia and/or septicemia (bacteria gets in the blood) or even death.
Most susceptible are persons with chronic health conditions and previous methicillin-resistant Staphylococcus aureus (MRSA) infections. In addition, people with open wounds, receive long term treatment and/or improper use of antibiotics, have invasive devices such as catheters or surgical drains, or have had prolonged or repeated hospital stays may also be at increased risk.
The main mode of transmission of VRSA is via hands. Transmission is also by direct contact with a person who has a draining skin lesion or wound.
Methicillin-resistant Staphylococcus aureus (MRSA) is a bacterium that causes infections in different parts of the body. It’s tougher to treat than most strains of staphylococcus aureus — or staph — because it’s resistant to some commonly used antibiotics.
The symptoms of MRSA depend on where you’re infected. Most often, it causes mild infections on the skin, like sores or boils. But it can also cause more serious skin infections or infect surgical wounds, the bloodstream, the lungs, or the urinary tract.
Though most MRSA infections aren’t serious, some can be life-threatening. Many public health experts are alarmed by the spread of tough strains of MRSA. Because it’s hard to treat, MRSA is sometimes called a “super bug.”
A large number of independent studies confirm copper’s antimicrobial effect, even against antibiotic-resistant bacteria such as MRSA (Methicillin-resistant Staphylococcus Aureus) and VRSA (Vancomycin-resistant Staphylococcus Aureus). The mechanisms of antimicrobial action by copper and its alloys, including brass, are a subject of intense and ongoing investigation.
- EPA registers copper-containing alloy products, May 2008
- Michel, James H., Moran, Wilton, R., Michels, Harold T., and Estelle, Adam A. (June 20, 2011). “Antimicrobial copper displaces stainless steel, germs for medical applications: Alloys have natural germ-killing properties”. Tube and Pipe Journal.
- Noyce, J.O.; Michels, H.; and Keevil, C.W. (2006). “Potential use of copper surfaces to reduce survival of epidemic methicillin-resistant Staphylococcus aureus in the healthcare environment” (PDF). Journal of Hospital Infection. 63 (3): 289–297. PMID 16650507. doi:10.1016/j.jhin.2005.12.008. Archived from the original (PDF) on 2012-01-17.
- Schmidt, MG (2011). “Copper surfaces in the ICU reduced the relative risk of acquiring an infection while hospitalized”. BMC Proceedings. 5: O53. PMC 3239467 Freely accessible. doi:10.1186/1753-6561-5-S6-O53.
- “TouchSurfaces Clinical Trials: Home”. coppertouchsurfaces.org.
- “355 Copper Alloys Now Approved by EPA as Antimicrobial”. Appliance Magazine. June 28, 2011.
- Kuhn, Phyllis J. (1983) Doorknobs: A Source of Nosocomial Infection? Archived February 16, 2012, at the Wayback Machine. Diagnostic Medicine
- Espίrito Santo, Christopher; Taudte, Nadine; Nies, Dietrich H.; and Grass, Gregor (2007). “Contribution of copper ion resistance to survival of Escherichia coli on metallic copper surfaces”. Applied and Environmental Microbiology. 74 (4): 977–86. PMC 2258564 Freely accessible. PMID 18156321. doi:10.1128/AEM.01938-07.
- Santo, C. E.; Lam, E. W.; Elowsky, C. G.; Quaranta, D.; Domaille, D. W.; Chang, C. J.; Grass, G. (2010). “Bacterial Killing by Dry Metallic Copper Surfaces”. Applied and Environmental Microbiology. 77 (3): 794–802. PMC 3028699 Freely accessible. PMID 21148701. doi:10.1128/AEM.01599-10.
Other Testing And Results
NON-GLP STUDY REPORT
STUDY TITLE
Ex-Vivo Antibacterial Evaluation of Topical Products Using a Vitro-Skin® Model
(ATCC 6538)
PRODUCT IDENTITY: C-220 Copper Sheet 0.050″ Thickness
AUTHOR – Matthew Sathe, B.S. Senior Microbiologist
STUDY COMPLETION DATE
October 27, 2017
PERFORMING LABORATORY
Accuratus Lab Services
1285 Corporate Center Drive, Suite 110
Eagan, MN 55121
STUDY REPORT
General Study Information
| Study Title: | Ex-Vivo Antibacterial Evaluation of Topical Products Using a Vitro-Skin® Model |
| Project Number: | A24209 |
| Protocol Number: | BOV002100917. EXVO |
Test Substance Identity
| Test Substance Name: | C-220 Copper Sheet 0.050″ thickness |
Study Dates
| Date Sample Received: | October 11, 2017 |
| Study Initation Date: | October 18, 2017 |
| Experimental Start Date: | October 24, 2017 |
| Experimental End Date: | October 26, 2017 |
| Study Completion Date: | October 27, 2017 |
The test organisms used in this study were obtained from the American Type Culture Collection (ATCC) Manassas, Va.
| Test Substance Preparation: | Ready to Use (RTU) copper bar |
| Test Exposure Time: | 1 minute (rubbing the bar on the inoculated and dried Vitro-Skin®) |
| Exposure Temperature: | Ambient (20° C with 28% relative humidity) |
| Number of Carriers Tested: | 2 tests and 2 control carriers |
| Organic Soil Load: | none |
| Neutralizer: | Letheen Broth (20 ml) |
| Agar Plate Medium: | Tryptic Soy Agar with 5% Sheep Blood |
Experimental Design
A film of the test organism dried onto a 1″ x 1″ demarcated area of 1.5″ x 1.5″ rehydrated Vitro-Skin® carriers was treated by rubbing the copper bar test material in circles, with light to medium pressure, over the inoculated surface for the exposure time. Following exposure, each carrier was neutralized and assayed for survivors. Appropriate culture purity, neutralizer sterility, carrier sterility and population controls were performed. Percent and Log10 reductions were determined for the test based on the test population control results.
Per Sponsor’s direction, the study was not required to be conducted under U.S. EPA 40 CFR Part 160 or U.S. FDA 21 CFR Part 58.
Table 2: Population Control Results
CFU = Colony Forming Unit
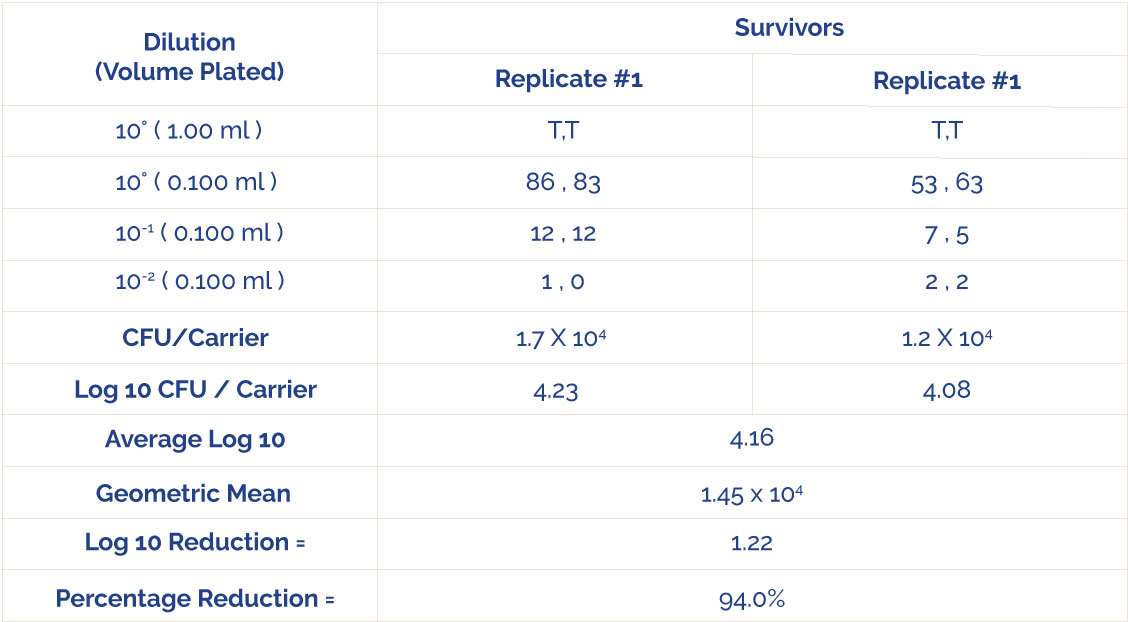
Table 3: Test Results Against
Staphylococcus aureus (ATCC6538)
CFU = Colony Forming Unit
T = Too Numerous To Count (>300)
Control Results
Analysis
C-220 Copper Sheet 0.050″ Thickness, ready to use, demonstrated a 94.0% (1.22 10910) reduction of Staphylococcus aureus (ATCC 6538) following a 1 minute rubbing exposure time at ambient temperature (20° C).
DOWNLOADS
Bacterial Killing by Dry
Metallic Copper Surfaces
Mechanisms of Contact
Mediated Killing of Yeast Cells on
Dry Metallic Copper Surfaces
DOWNLOADS
Bacterial Killing by Dry
Metallic Copper Surfaces
Mechanisms of Contact
Mediated Killing of Yeast Cells on
Dry Metallic Copper Surfaces
CopperTouch, LLC © 2021. All Rights Reserved |
CopperTouch, LLC © 2021. All Rights Reserved |